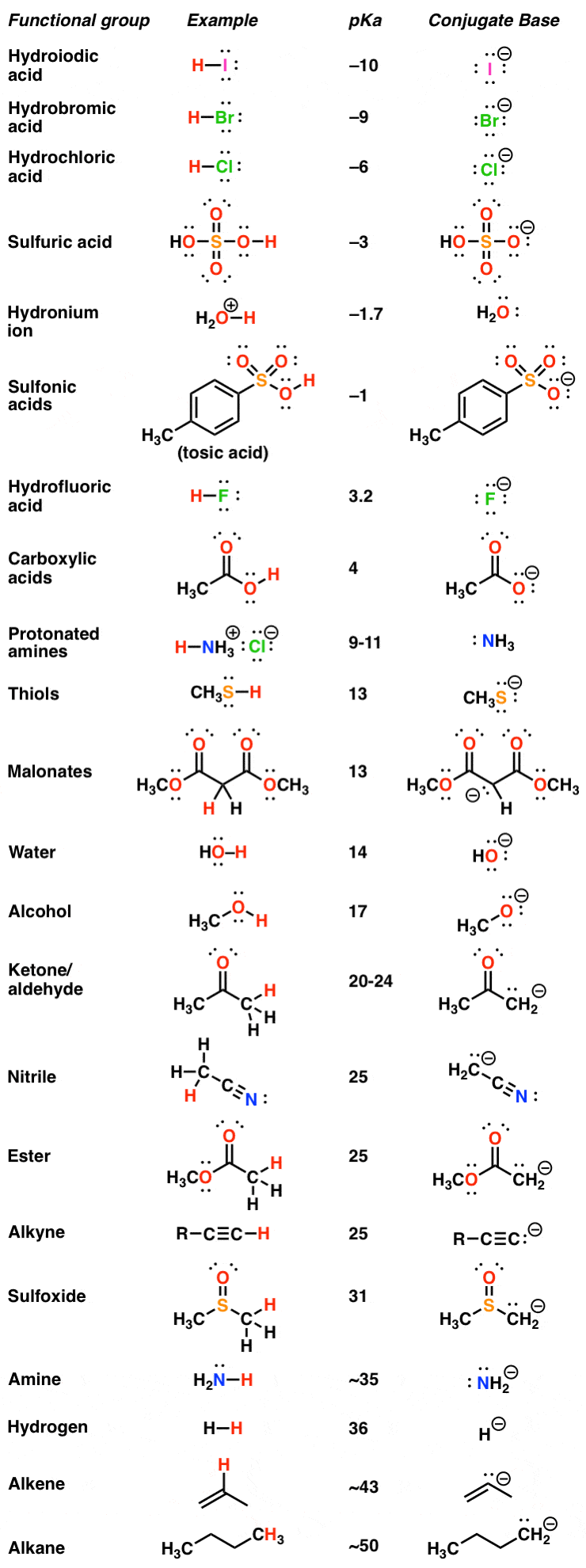
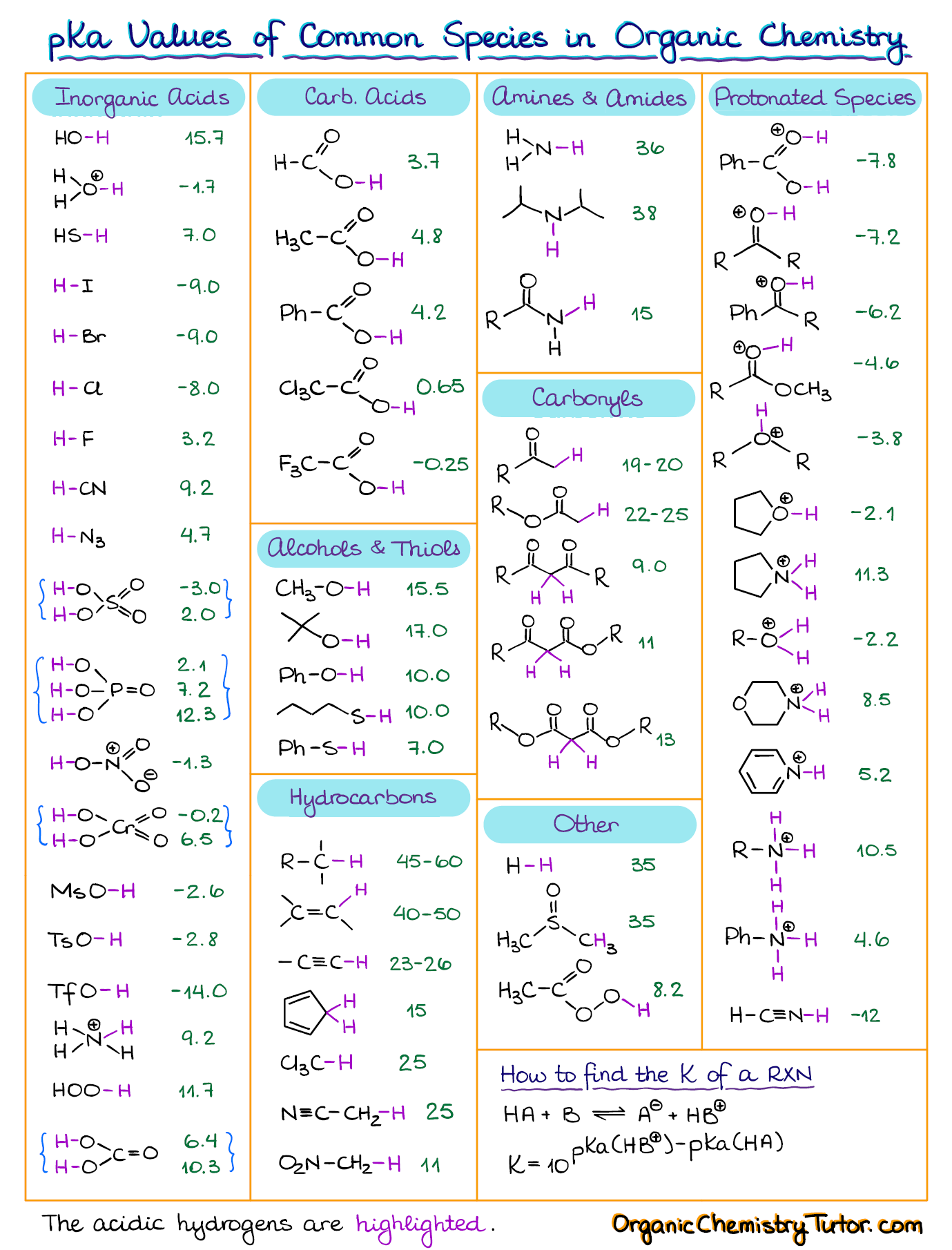
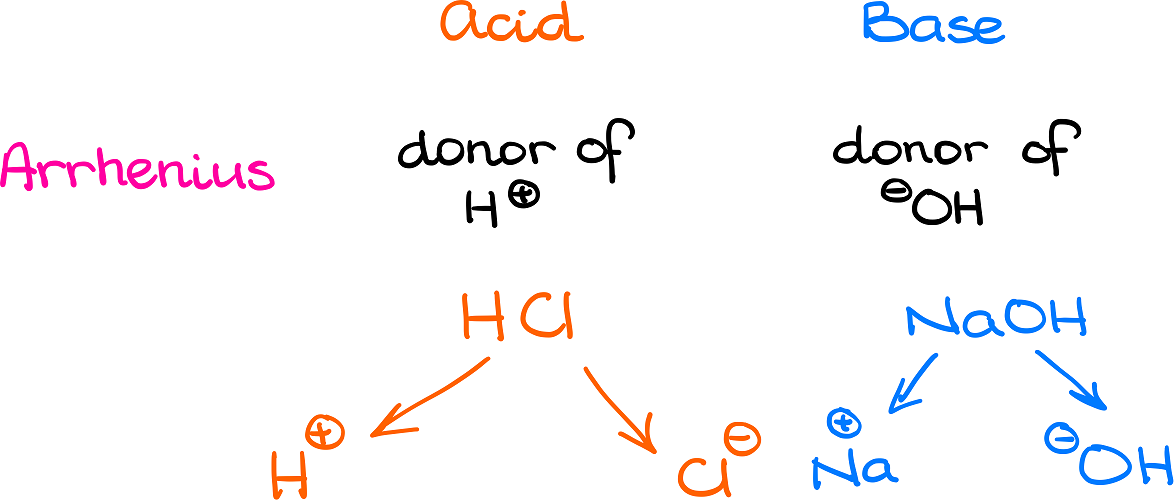
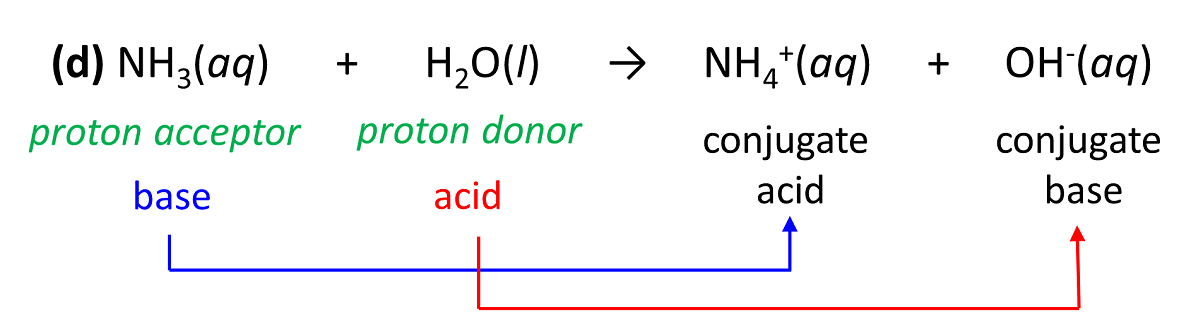
3.3: pKa of Organic Acids and Application of pKa to Predict Acid-Base Reaction Outcome - Chemistry LibreTexts

It has been found that the pH of a 0.01 M solution of an organic acid is 4.15 . Calculate the concentration of the anion, the ionization constant of the acid and its pKa .
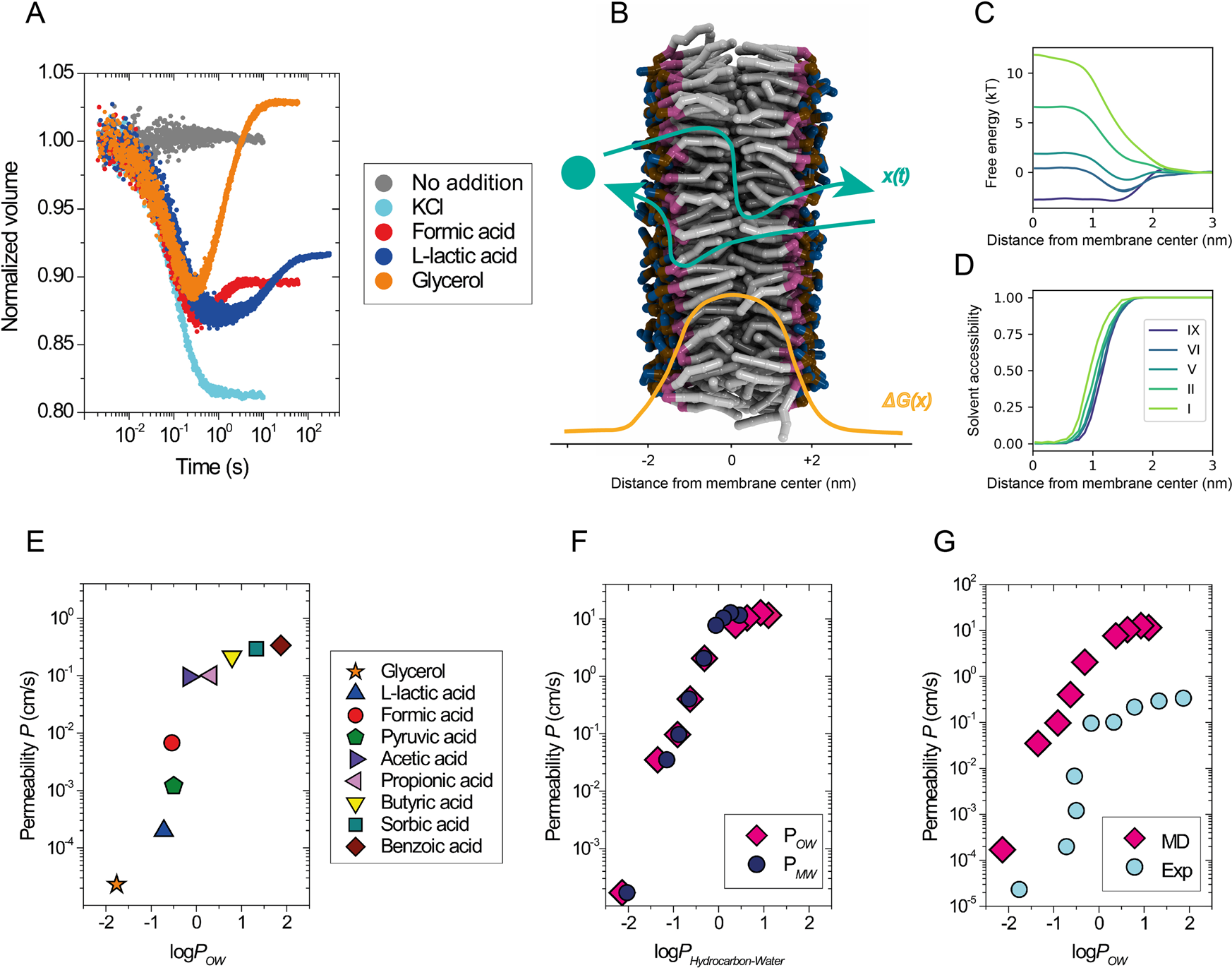
Membrane thickness, lipid phase and sterol type are determining factors in the permeability of membranes to small solutes | Nature Communications